Which Best Describes the Shape of the Water Molecule
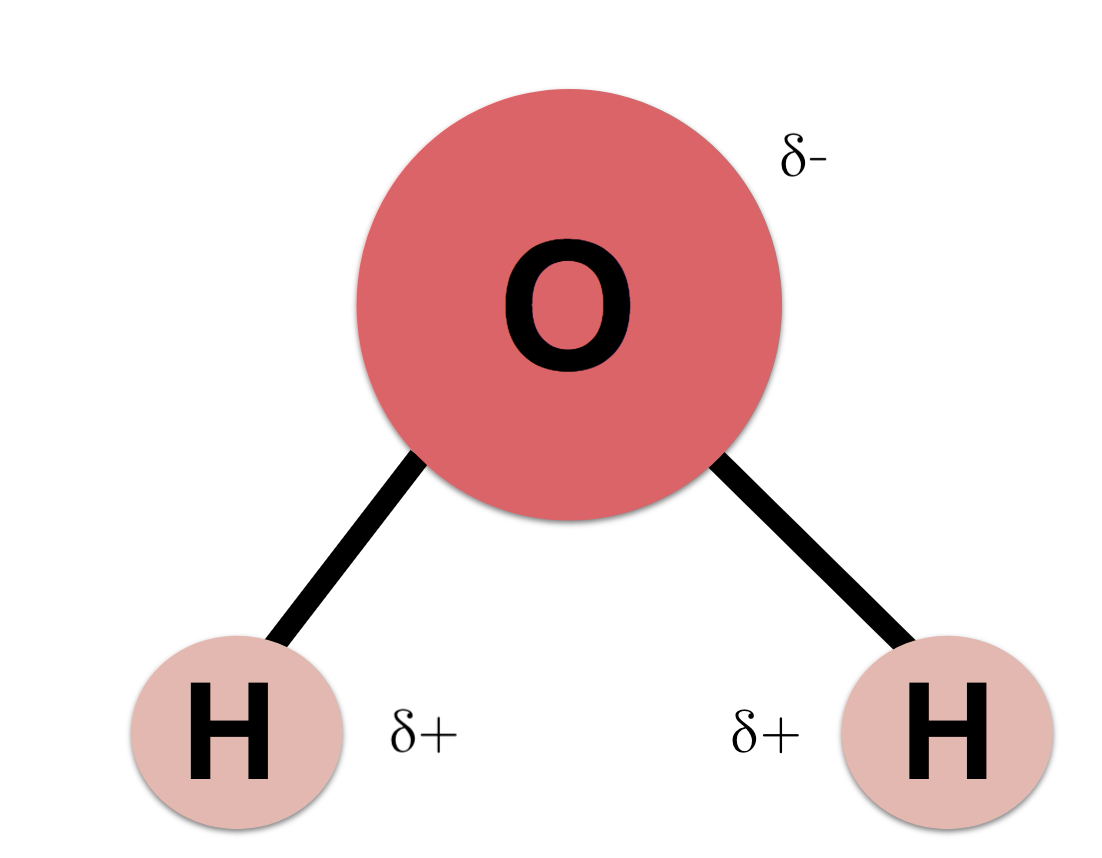
Water H 2 O is polar because of the bent shape of the molecule. You can add the two remaining valence electrons onto O and check that.
Why Life Depends On Water Biology For Non Majors I
Q7 Water molecule has 2 bonds and 2 lone pairs around the central atom oxygen therefore its molecular shape is bent.
. Which best describes the size and shape of a sample of gas. There are four electron domains in a water molecule. You cannot smell as well when you have a cold.
It has a definite volume but shape is determined by the container E. What shape would a molecule of hydrogen sulfide have. Two domains are bonding pairs and the other two domains are lone pairs of electrons.
The central oxygen atom has 2 non-bonding pairs of electrons and 2 pairs of shared bonding electrons each to a hydrogen atom. A hydrogen sulfide molecule H2S has a bent shape similar to that of a water molecule. The electronegativity value of hydrogen is 21 while the electronegativity of oxygen is 35.
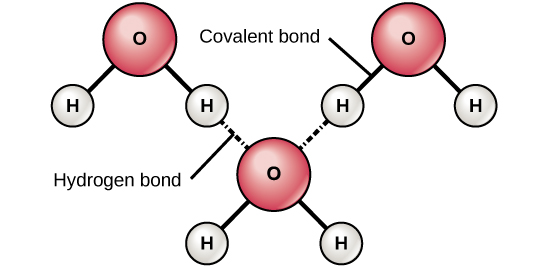
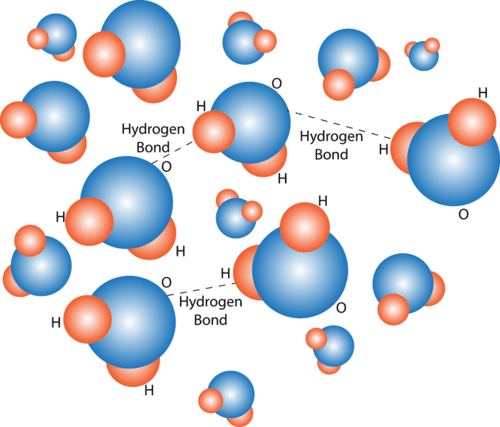
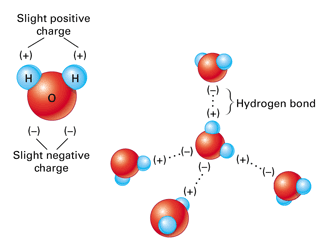
Consequently the electrons in the water molecule spend slightly more time around the oxygen atomic center and less time around the hydrogen atomic. Water is a unique molecule that is essential for life as we know it. Hydrogen bonding occurs when the negative dipoles of the.
Start your trial now. The hydrogen sulfide H2S molecule has a bent shape. Oxygen has six valence electrons but as it tends to accept two it tends to make two bonds in covalent compounds.
What phrase best describes the arrangement of these electron groups around the central oxygen atom. The molecule can have the same or different types of atoms. The lone pairs repel the bonding pairs to a bent shape.
The answer is option C Q8 The molecules that contains N-H O-H or F-H bonds can form hydrogen bonds which is the strong intermolecular attraction force. One electron group means one lone pair one single bond one double bond or one triple bond. A water molecule consists of two atoms of hydrogen linked by covalent bonds to the same atom of oxygen.
3 This molecule is essential in the life of living beings serving as a medium for the metabolism of biomolecules is found in nature in its three states and was key to its formation. The reason the shape of the molecule isnt linear and nonpolar eg like CO 2 is because of the difference in electronegativity between hydrogen and oxygen. Which best describes a molecule.
Eventually you should get that formaldehyde looks like this. Volume and shape cannot be described. Water H2O is an inorganic chemical compound formed by two hydrogen H and one oxygen O atoms.
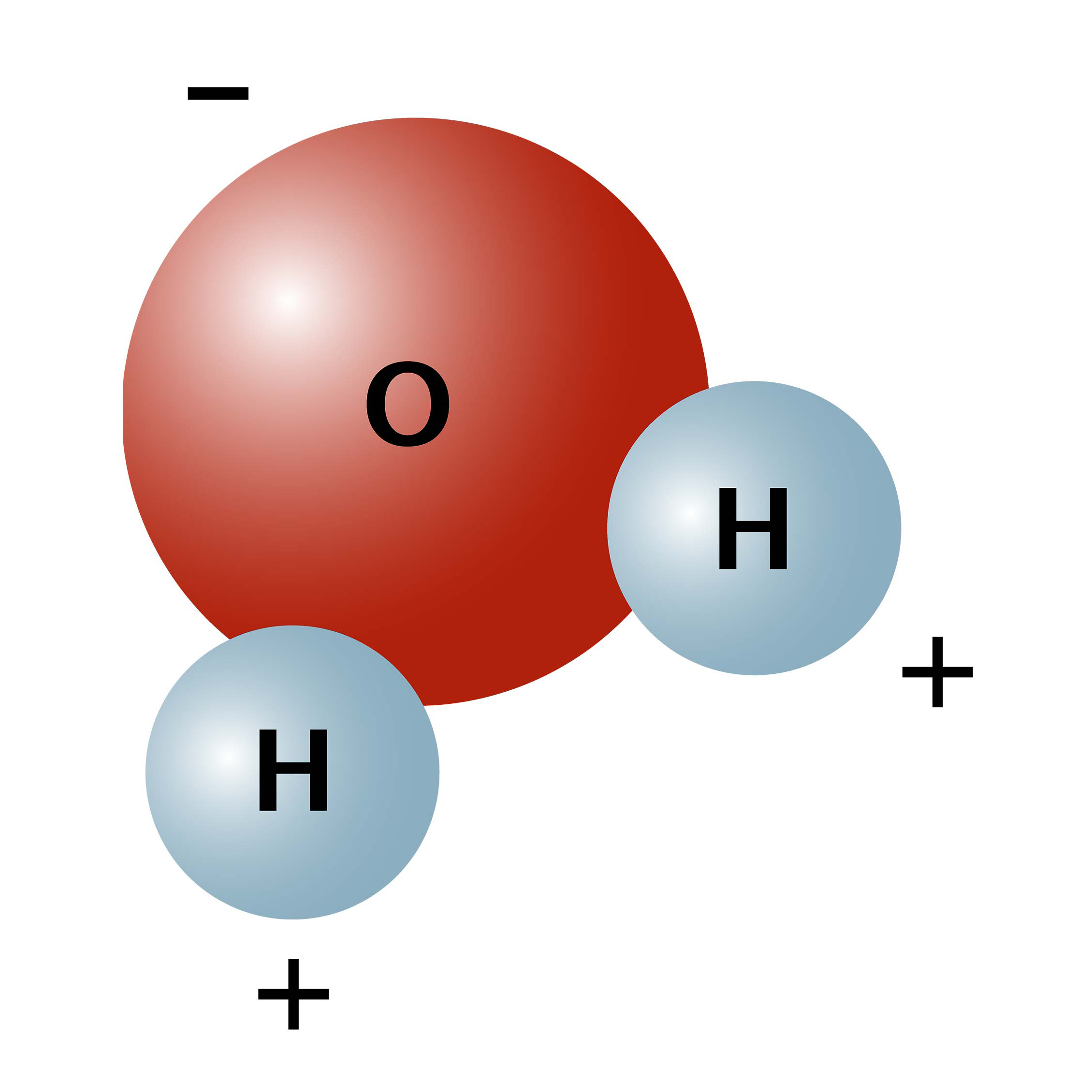
Hydrogen is more electronegative than oxygen causing a partial negative charge on hydrogen and a. What word or two-word phrase best describes the shape of the water H0 molecule. Molecular geometry is the three-dimensional arrangement of the atoms that constitute a moleculeIt includes the general shape of the molecule as well as bond lengths bond angles torsional angles and any other geometrical parameters that determine the position of each atom.
It is necessary to distinguish between drinking water. Solution for What word or two-word phrase best describes the shape of the water H0 molecule. The shape of water molecule is asymmetrical.
However because the two non-bonding pairs remain closer to the oxygen atom these exert a stronger repulsion against the. The reason for many of waters properties is due to hydrogen bonding. Its volume is determined by the container but it has a definite shape D.
Hydrogen is almost always going to make only one bond given that it only has one valence electron. The water molecule may result in a tetrahedral geometry in which the angle between electron pairs and therefore the H-O-H bond angle is 104. It has definite volume and definite shape C.
Hydrogen is highly reactive. What word or two-word phrase best describes the shape of the. Molecular geometry influences several properties of a substance including its reactivity.
How many electron groups are around the central oxygen atom. Which best describes the reason why a water molecule has a bent shape. - 13348791 A molecule is 2 or more atoms bonded together.
Volume and shape are both determined by the container B. Which describes the structure of a water molecule H2o. Answer the questions in the table below about the shape of the water H20 molecule.
A water molecule has a v shape with two covalent bonds spread apart at a 1045 o angle. It has two polar single bonds and a bent shape. Atoms of oxygen are electronegative and attract the shared electrons in their covalent bonds.
First week only 499. The arrangement of the electron pairs around the central oxygen is tetrahedral with the non-bonding pairs taking up slightly more space than the bonding pairs but since we dont see the the electrons but only the atoms the molecule.

The Strong Polar Bond Between Water Molecules Creates Water Cohesion U S Geological Survey
Biochemical Properties Of Water Advanced Read Biology Ck 12 Foundation
Answer In General Chemistry For Jude 101935

Molecular Geometry Of A Water Molecule The Molecular Shape Is An Download Scientific Diagram

The Chemistry Of Water Water Molecules Nsf National Science Foundation

What Is The State Of One Molecule Of Water Under Standard Conditions Quora
Biochemical Properties Of Water Advanced Read Biology Ck 12 Foundation
Lesson Summary Water And Life Article Khan Academy

Hydrogen Bonding Read Chemistry Ck 12 Foundation
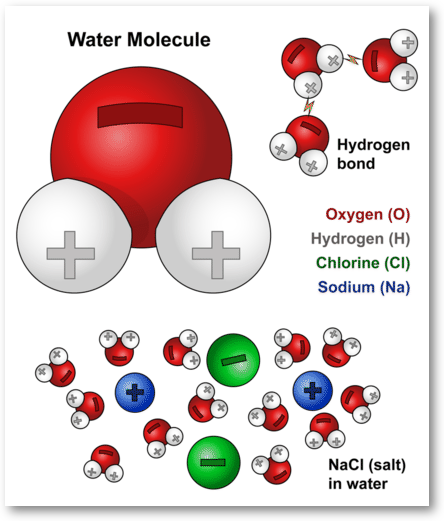
Water Molecules And Their Interaction With Salt U S Geological Survey

Soil Water From Molecular Structure To Behavior Learn Science At Scitable

The Structure And Properties Of Water Introduction To Chemistry

Describing Water Molecules Which Of These Statements Would Be True If The Water Molecule Was Linear Brainly Com

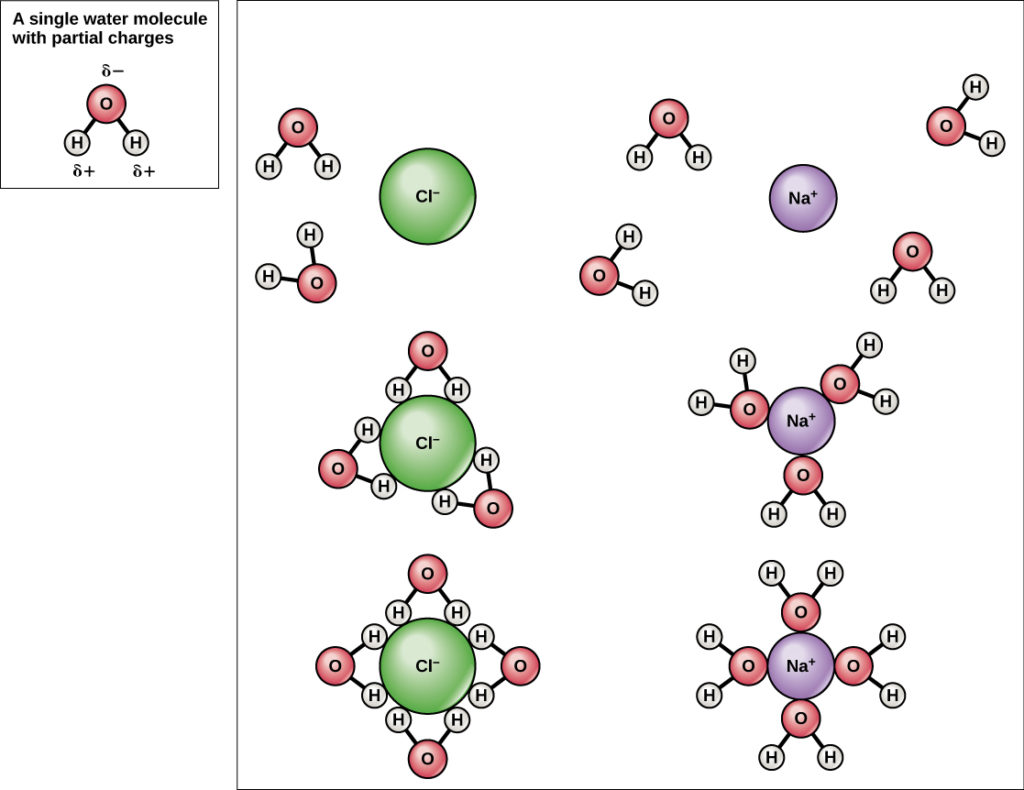
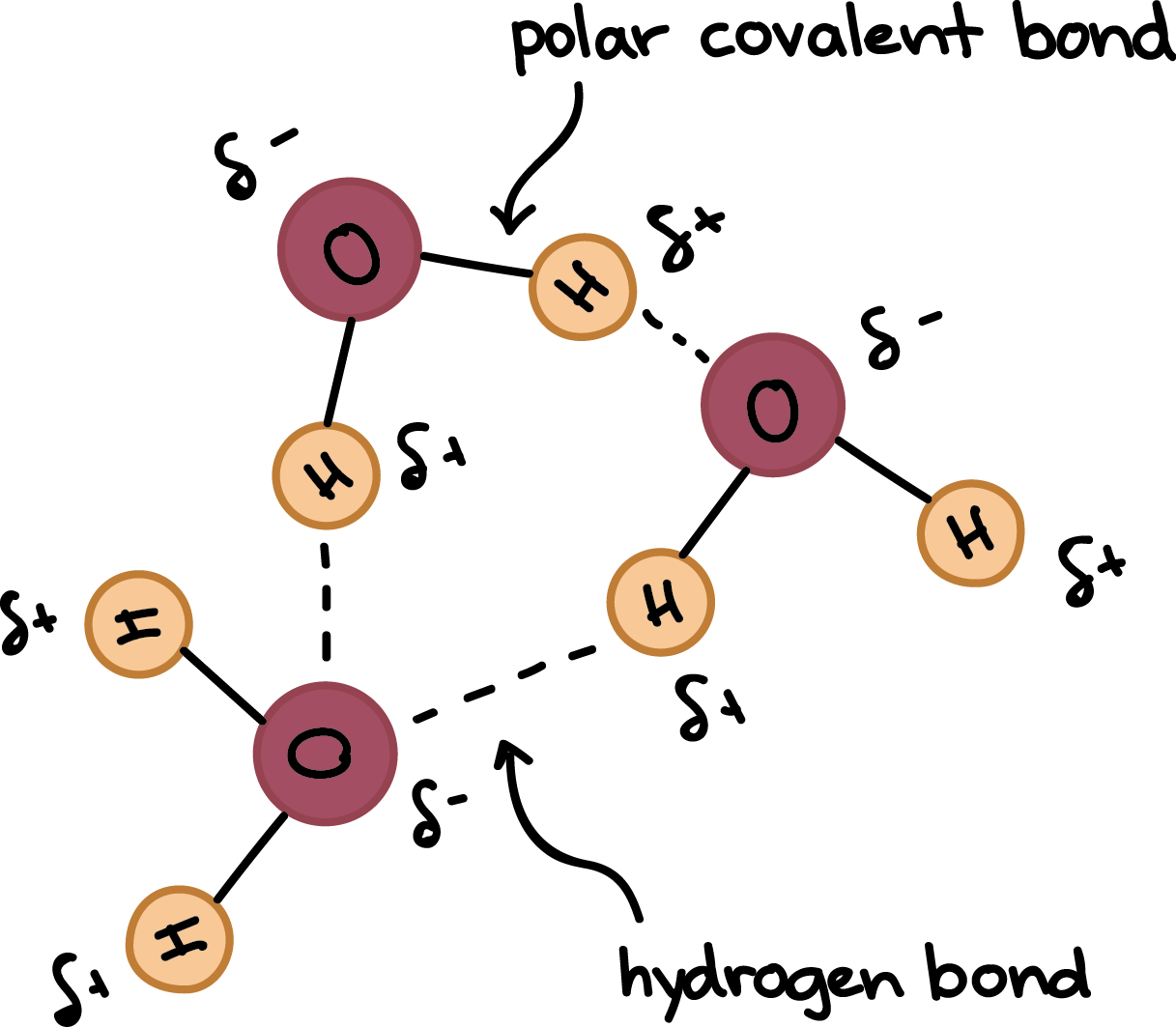
Comments
Post a Comment